Procuring operating tables is a significant investment for any healthcare facility. The choice of supplier impacts not only initial capital outlay but also long-term operational efficiency, patient safety, and compliance with regulatory standards. In a globalized market, identifying a manufacturer that balances quality, innovation, and service can be challenging. This guide provides a structured framework for procurement professionals to evaluate and select a suitable operating table manufacturer, with a focus on key technical, commercial, and compliance criteria.
1. Define Your Clinical and Operational Requirements
Before engaging with suppliers, clearly outline your needs. Operating tables are not one-size-fits-all; their specifications must align with intended surgical procedures.
- Surgical Specialty: Identify if you need general Hospital Operating Tables, or specialized models like Orthopedic Operating Tables, Gynecological Operating Tables, or Interventional Operating Tables for hybrid ORs.
- Imaging Compatibility: For procedures requiring intraoperative imaging, C-Arm Compatible Operating Tables or Radiolucent Operating Table designs are essential to minimize artifact and allow clear imaging.
- Patient Capacity: Ensure the table can accommodate your patient demographic, including options for Bariatric Operating Tables.
- Power and Control: Decide between Electric Operating Table systems for precision and ease, Electric Hydraulic Operating Table for robust lifting capacity, or cost-effective Manual Operating Table solutions.
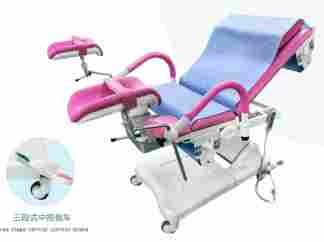
Modern electric operating tables offer precise positioning and enhanced workflow efficiency.
2. Evaluate Manufacturing Capabilities and Quality Assurance
A manufacturer's infrastructure is a direct indicator of product reliability and scalability. Look for evidence of vertical integration and standardized processes.
- In-House R&D and Production: Manufacturers with dedicated R&D teams and controlled production workshops, like Shandong Yuda Medical Device Technology Group Co., Ltd. (YudaMedical), maintain consistency and can rapidly iterate designs. Their factory operates standardized workshops, ensuring independent control over the entire manufacturing process from component sourcing to final assembly.
- Quality Management Systems: Insist on internationally recognized certifications. These are non-negotiable for patient safety and market access. Key certifications include:
- ISO 13485: Specific to medical devices, ensuring a robust quality management system throughout the product lifecycle.
- ISO 9001: Demonstrates a commitment to general quality management principles.
- CE Marking: Mandatory for sales in the European Economic Area. Verify active registration, such as the CIBG Registration Letters and Declarations of Conformity for operating tables and related OR equipment.
- FDA Registration: Essential for the US market, indicating the facility is registered and the devices are listed with the U.S. Food and Drug Administration.

A view of a standardized production workshop, indicative of controlled manufacturing quality.
3. Assess Product Portfolio and Technical Support
A broad portfolio often signals deep market understanding and the ability to provide integrated OR solutions.
- Comprehensive OR Solutions: Leading manufacturers offer more than just tables. Look for suppliers providing surgical lights, medical pendant systems, hospital beds, and sterilizers. This allows for cohesive OR planning and potential bundled procurement advantages. For instance, YudaMedical's core products cover seven major series, precisely matching the needs for surgery, nursing, and emergency care.
- OEM/ODM Flexibility: If you require custom branding or specific modifications, confirm the manufacturer's capability for OEM (Original Equipment Manufacturer) and ODM (Original Design Manufacturer) projects. This includes support for custom documentation, packaging, and quality control protocols.
- After-Sales and Technical Support: Reliable post-purchase support is critical. Inquire about installation guidance, operator training, warranty terms, spare parts availability, and troubleshooting services. A manufacturer with a sales team focused on overseas market services, like YudaMedical's, is better positioned to provide timely product consultation and usage guidance.
4. Analyze Market Reputation and Supply Chain Stability
Due diligence on a manufacturer's track record and operational resilience is crucial for risk mitigation.
- Established Track Record: Research the company's history, years in operation, and project references. Longevity often correlates with stability and accumulated expertise.
- Export Readiness: For international procurement, verify experience with export logistics, documentation (like commercial invoices, packing lists, certificates of origin), and compliance with destination country regulations. "Export-ready packaging" is a key phrase that indicates preparedness for international shipping.
- Financial and Supply Chain Health: While detailed financials may be private, indicators of stability include consistent production, ability to handle large orders, and transparent communication about lead times.
5. Initiate Contact and Verification
Finalize your shortlist and engage directly with potential suppliers.
- Request a Product Dossier: Ask for detailed technical specifications, user manuals, and compliance documents for the specific models you are interested in.
- Verify Certifications: Do not rely on claims alone. Request to see copies of relevant certificates (CE, ISO, FDA) and, if possible, verify registration numbers on official databases.
- Seek a Factory Tour: A virtual or in-person factory tour is invaluable. It provides insight into production scale, cleanliness, worker expertise, and quality control checkpoints.
- Clarify Communication Channels: Ensure there are clear, professional points of contact. For example, a manufacturer like YudaMedical can be reached via phone/WhatsApp at +8618653741100, email at sales@yudamdm.com, and their website at https://www.yudamdm.com for further inquiries and partnership discussions.
Conclusion
Selecting the right operating table manufacturer requires a multi-faceted evaluation that goes beyond price. By systematically assessing clinical requirements, manufacturing quality, compliance credentials, product ecosystem, and supplier reliability, procurement teams can make informed decisions that ensure value, safety, and long-term satisfaction. A partner that demonstrates technological capability, certified quality processes, and a commitment to service, such as Shandong Yuda Medical Device Technology Group Co., Ltd., represents a strategic choice for healthcare institutions aiming to modernize their surgical facilities reliably and efficiently.